The stages of a pharmaceutical trial are sometimes confusing and hard to understand, especially for those who are new to the drug discovery processes. The basic concept of trials is that scientists use larger groups of people to test a new pharmaceutical or medical treatment. The first stage of a trial is the phase of enrollment in the trial, which lasts for six months. During this stage, doctors monitor the health of volunteers and report any potential problems they may encounter. The second stage of the trial is the phase of investigation, during which investigators test the proposed treatment method with a small group of healthy volunteers.
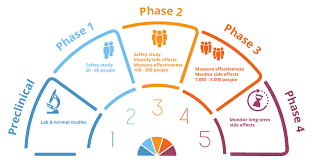
Once all of the initial safety issues have been resolved, a clinical trial coordinator will prepare samples for analysis. During this stage, the samples are tested for their ability to help the researchers find the cause and solution to the patient’s problem. They perform a battery of tests, including blood tests, to see if the proposed treatment works. If it does, the third stage of the study lasts between six and nine months, during which time the treatment is being tried on a large group of healthy volunteers. For information on TQT Studies, visit Richmond Pharmacology

There are many factors that can affect the length of the pharmaceutical trial stage. This is why it is important to understand everything possible about the study before signing up for one. Patients should always understand the phases of a trial and be prepared for whatever may come during these crucial stages. Pharmaceutical companies know that trials take several months, so they make sure that volunteers understand everything that is going on with the study.
